|
The class reviewed the Ohm's Law and Electrical Current Problems from last week.
Please hand these two worksheets in! The following notes were taken and the two worksheets were worked on.
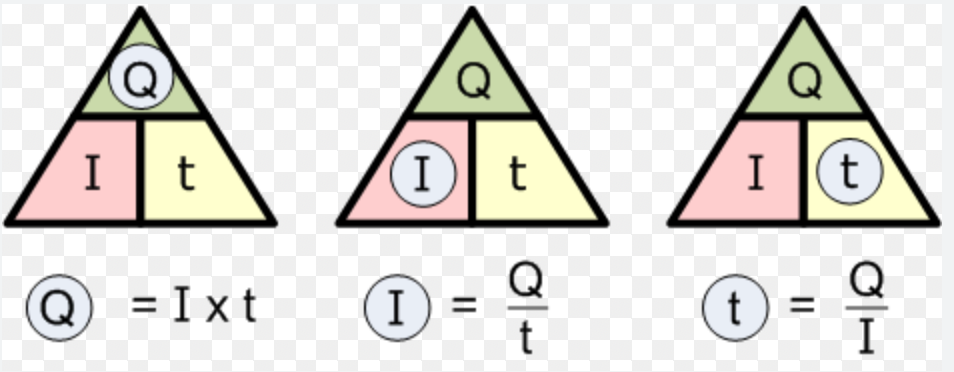
Notes... The symbols for a voltmeter, ammeter and resistor were added to the last physics notes and Table 1 on page 322 was copied into notes. Calculating Current I=current (amperes, A) t=time (seconds, sec) Q=charge (coulombs, C) 1C=6.25x10^18 electrons Examples...
Please work on these worksheets at home and bring any questions you have to class tomorrow! An attendance question was done to start the class. Next we did a white boarding activity answering the questions below... then we took the following physics notes. Pages 302-304 and 315-317 in the textbook were read out-loud and then we worked on making calculations for circuits with different V/I/R.
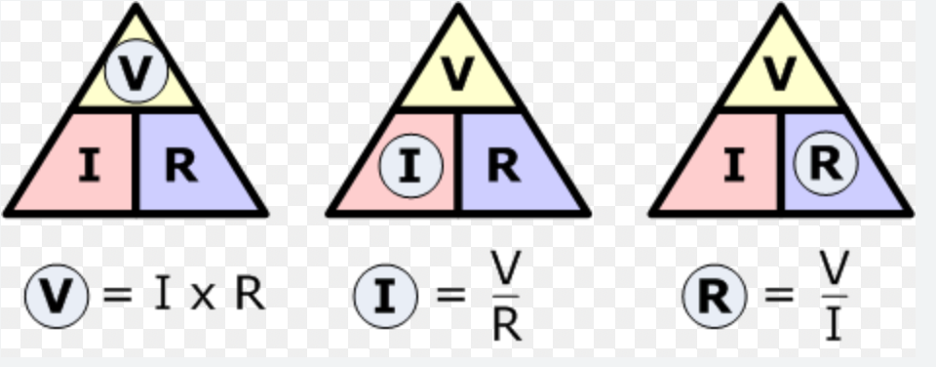
-What is physics? -What is voltage, resistance and current? -Why do batteries have a + and - side? -How can electricity be dangerous? Notes... -Physics is one of the most fundamental scientific disciplines, with its main goal being to understand how the universe behaves. Natural science that studies matter, motion and behavior through space and time, energy and force. V -When an electron has the ability to move in a circuit, it has potential energy. Voltage is the difference in potential energy between two points in a circuit. Volts, V. I -A voltage source pushes electrons through a circuit creating a current (the flow of electrons). Measured in Amperes/Amps, A. R -A load (like a light bulb) slows down these electrons to transform their energy into another form (light and heat). This slowing of electrons is called resistance. Measured in ohms, Ω. -Electrons flow from the negative terminal of a battery to the positive terminal of a battery. -Table 1 on page 304 was copied out with the addition of resistors. The class explored electrical circuits and tested the equipment to prepare for the physics unit starting after spring break.
I hope you all have a lovely break :) The class marked their chemistry tests. Then we exploded sodium, completed the elephant toothpaste lab, and spent the last 15mins outside doing an exit question and talking about the next units we have coming up in science 9.
Today the amazing class TA did a lesson on chemical and physical changes. After, students were given time to work on the chemistry review and ask questions about the test tomorrow!
Today we had a short quiz on Bohr models and naming compounds. After, students worked on their chemistry review package. Students also filled out a chemistry pre test reflection.
There will be a chemistry test on Tuesday next week! Please bring your completed review to class and hand it in because it will count as marks towards your test! An attendance question was done to start the class today and then we reviewed covalent compounds as a class. After, we learned how to name Ionic Compounds and read pages 245-246 in the textbook.
Please finish the 'Naming Covalent and Ionic Compounds' worksheets for Friday! We will also have a small naming quiz on Friday. Today we learned how to name covalent compounds.
Please finish the worksheet below and lets go over these questions in class tomorrow! Today we did an outside element scavenger hunt in groups!
Element Scavenger Hunt Rules... -photos must be taken outside -you must email your photos, group members names, and list the elements you found by the end of class -extra points awarded if all three of your groups members are in the photos -extra points awarded if your photos are of pure elements Winning group and runner up will be announced tomorrow in class! (prizes!!) Today the class started with an attendance question and then worked on their 'Element Inquiry Project' on Teams. After break, the students did their grade 10 course selection and then continued working on their projects.
Please hand in your 'Element Inquiry Project' on Teams and bring your completed Bohr drawings to class on Friday! Today we learned about Bohr Models.
Please come see me if you were not here this day :) The class worked on their 'Element Inquiry Project' on Teams. Please complete this and hand it in for homework! Today the class watched the following videos on leptons, quarks, and plasma. After, we went over the families in the periodic table and then did the PHET games in the Teams assignment - try to get five stars on all the games!
New words? Ductile - can be drawn out into a thin wire Malleable - can be made into sheets Lustre - shiny! Please do the PHET games for homework if you did not get them done in class! The class started off with an attendance question and then watched the following videos and did the following notes. We also colored in our periodic table as follows, looked at the scale of the universe, and completed the 'Build an Atom Activity' on Teams! Notes... Matter - physical substance (liquid, solid, gas) Particle - matter that has multiple atoms in it Atom - basic unit of chemical elements Ex. Carbon atom Element - cannot be broken down into another substance Molecule - groups of atoms bonded together Ex. N2 and H2O Compound - two or more separate elements bonded together Ex. H2O Subatomic Particle - particles that are smaller than an atom Protons - (positive charge) and neutrons (zero charge) are made of quarks are held together in the nucleus by a strong nuclear force. Electrons - (negative charge) are a type of lepton and are held at a distance from the nucleus through electromagnetism. Please hand in your 'Build an Atom Activity' on Teams'! The class completed the following density lab and answered the two analyze questions to go along with it.
Please come see me if you were not here this day! PLEASE BRING A POP CAN TO SCHOOL TOMORROW :) The class started off with an attendance question and then moved onto reviewing grade 8 chemistry! The following notes were taken and the density worksheet was worked on. After, we did the following lab and handed in questions 1-5 on it.
Notes... *State changes triangle + solid, liquid, gas drawings Kinetic Molecular Theory -All matter is made up of tiny particles -Different substances have different particles -The particles are in constant motion -The more energy the particles have, the faster they move -There is empty space between the particles Atomic Molecular Theory -All matter is composed of small, fast moving particles called atoms. These atoms can join together to form molecules. This theory contains many other theories that explain chemistry, and nature, and everything?! -Density is a measurement of how much mass fits in a given volume. -Mass is a measurement of how much matter (p,n,e) there is in the object. -Volume is a measurement of how much 3D space the matter occupies. -Density is calculated by dividing the mass of an object by its volume 𝐷𝑒𝑛𝑠𝑖𝑡𝑦= 𝑀𝑎𝑠𝑠/𝑉𝑜𝑙𝑢𝑚𝑒 Please complete the following density questions and then them in and come see me if you were not here this day to complete the lab! The class worked on an excel graphing assignment on Teams.
Please hand this in and come see me if you were not here this day! The class continued working on their lab reports.
Please hand your lab reports and data sheets in! The class recorded their data on their gummy bears and then worked on their lab report, the assignment is on Teams.
The class did a quick attendance question and then tested their 'Paper Bridges Experiment'. After, they did lab safety and then did the Gummy Bear Lab.
Please come see me if you were not here this day and complete the lab safety if you did not in class! The class was put into random groups and then did the paper bridges experiment!
|
hello!Thank you for taking the time to commit to your version of success in this course - please email me if you have any questions. Archives
June 2023
|